Science-Driven.
Risk-Controlled.
Designed for Robustness.
ValGenesis iCMC™ enables quality by design in pharma with intelligent frameworks that drive robust processes, consistent product quality, and regulatory alignment.
You Will be in Great Company
Join the life sciences leaders using the ValGenesis Process Lifecycle Suite to remove barriers to innovation and accelerate delivery—by making process lifecycle execution consistent, connected, and efficient.



Customer Testimonials

When we decided to go with iCMC, we wanted to have the roll-out concluded within six months. The support we received from the team, their willingness to accommodate our requests and timelines, and their expertise in resolving all the issues that came up during the project was amazing.
Dr. Iris Ziegler
Director QbD & Pharmaceutical Sciences

We work with ValGenesis iCMC to digitalize our QbD framework within GSK for the development of new vaccines. We are confident that iCMC will be key for simplification, acceleration, and knowledge management.
Hervé Gressard
Head of Global Project Statistics CMC Technical R&D
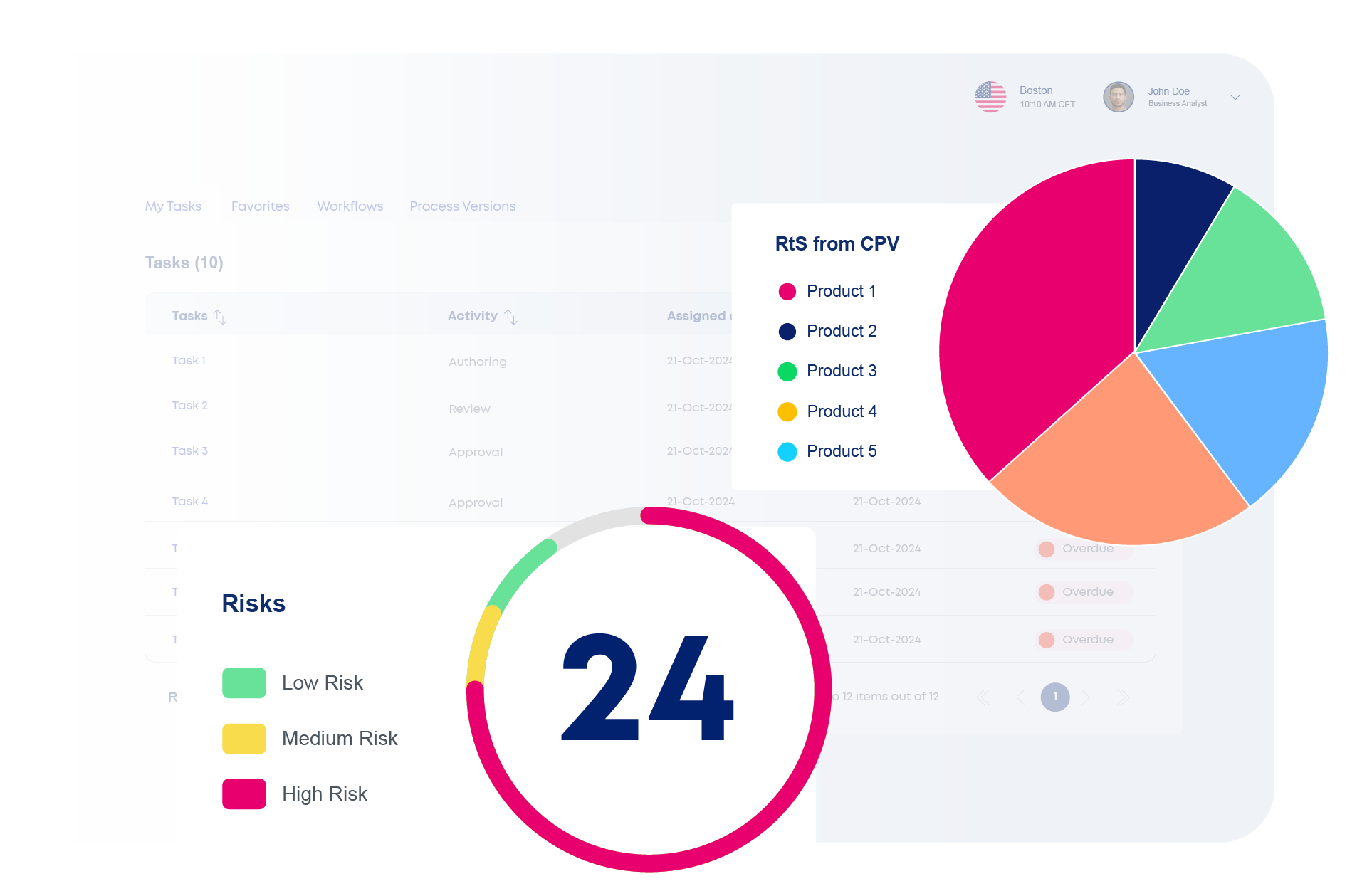
Discover How iCMC works as part of the ValGenesis Process Lifecycle Suite
iCMC centralizes process and analytical development—supporting QTPP definition, CQA and CPP analysis, and control strategy design in a collaborative, traceable environment. Teams can apply industry-standard risk assessment tools for pharmaceutical processes such as FMEA, PHA, HAZOP, and Ishikawa diagrams, with built-in guidance for regulatory alignment.
Advanced analytics, including predictive insights powered by machine learning, help teams identify trends, flag potential issues early, and optimize development strategies.
The platform supports digital validation planning, analytical method validation, project coordination, structured reporting, and knowledge reuse across products and facilities. With integration capabilities for QMS, MES, LIMS, and ERP, iCMC provides full lifecycle traceability—from early development to commercialization.
Overview
Design robust, science-based processes
Apply QbD and risk-based principles to build quality into every stage of CMC in drug development.
Ensure consistency across products and sites
Unify process knowledge and reduce variability through standardized, traceable workflows.
Meet regulatory expectations with confidence
Align with global guidelines and maintain audit-ready documentation at every step.
Resources

Integrating Digital Transformation and Quality by Design for Enhanced Pharmaceutical Development
Discover how digital platforms paired with QbD speed pharma development and support ICH compliance. ...
Read
Removing Bias from Risk Assessment
A data-driven, digital CMC development framework aligned that uses CPV evidence to reduce bias in ...
Read
Transforming Pharmaceutical Development: Combining Digital Platforms and QbD Principles
Cut paper bottlenecks with digital QbD workflows to speed risk assessments, reporting, and ...
Read
The Toolbox for an effective Tech Transfer
Speed up pharma tech transfer with a practical Pharma 4.0 toolbox for data, risk, and control ...
ReadWebinars

Reimagining Tech Transfers: How Automation and AI Assessments Deliver Smoother Processes
Tech transfer has always been a critical but challenging step in the pharma and biotech product ...
Watch
Revolutionizing CMC: Cutting Errors and Delays in Quality by Design
Quality by Design can transform how pharma organizations develop and manufacture products. But ...
Watch
Revolutionizing CMC in Drug Development: How to Reduce Errors and Delays in Quality by Design
Quality by Design in pharma offers significant potential—but many organizations struggle to ...
WatchVideos

Tech Transfer Inefficiency: Where CMC Timelines Bleed Cost
Cut CMC tech transfer delays by turning process knowledge into traceable digital flow. Watch now.
Watch
ValGenesis iCMC™: Science-Driven. Risk-Controlled. Designed for Robustness.
Replace scattered CMC docs with QbD-based, risk-controlled workflows in ValGenesis iCMC—watch now ...
Watch
Are You Still Doing QbD the Hard Way? The High Cost of Manual Data Entry
Cut manual QbD data entry in CMC. See how scattered data drives delays and errors—and how ...
Watch
The 3 Types of Quality Risk Management Tools and What They Do Best
Learn how identification, analysis, and evaluation tools support effective QRM. Understand how to ...
Watch
Don’t Let Spreadsheets Hold Back Your CMC: How ValGenesis Process Lifecycle Suite Transforms QbD Efficiency
Move CMC development beyond spreadsheets. See how a digital QbD platform structures risk, connects ...
WatchFrequently Asked Questions
From Molecule to Market. Faster.
Book a demo to see how ValGenesis iCMC™ can help you accelerate time to market.