Proactive Monitoring.
Real-Time Insights.
Quality Assured.
ValGenesis iCPV™ transforms continued process verification through automated data analysis for early issue detection and consistent product quality across sites and batches.
Your Manufacturing Will Be in Good Company
Join the life sciences leaders using the ValGenesis Process Lifecycle Suite to remove barriers to innovation and accelerate delivery - by making process lifecycle execution consistent, connected, and efficient.



Customer Testimonials

Implementing ValGenesis iCPV has transformed our approach to CPV. The integration of our data systems and the comprehensive training from the ValGenesis team have empowered our users, ensuring we meet regulatory standards while enhancing efficiency and data integrity.
Patricia Watanabe
Validation Supervisor
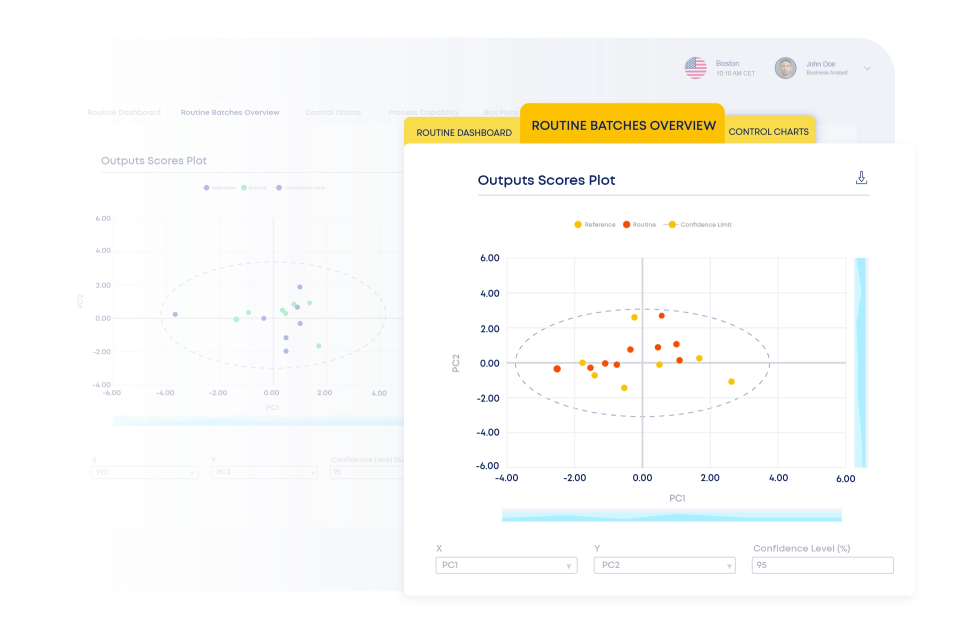
Discover How iCPV works as Part of the ValGenesis Process Lifecycle Suite
iCPV centralizes and automates continued process verification using multivariate statistical tools, control charts within a statistical process control (SPC) framework, and flexible, guided workflows.
From monitoring CPPs and CQAs to triggering alerts on process drift, iCPV ensures quality decisions are made in real time through multivariate and predictive data analytics in the pharmaceutical industry. Teams follow guided workflows to design, execute, and maintain CPV programs with confidence, supported by custom plots, automated updates, and standardized, automated reporting.
Integrated with LIMS, MES, SAP, and manufacturing systems, iCPV turns fragmented process data into actionable insights—delivering intelligent, compliant pharmaceutical manufacturing software for modern operations.
Overview
See issues before they happen
Detect trends early with multivariate analytics and real-time alerts to prevent deviations.
Streamline CPV from end to end
Automate report generation and minimize manual effort with guided, validated workflows.
Ensure continuous quality and compliance
Monitor CPPs and CQAs at scale, maintain audit readiness, and support regulatory assurance.
Resources

Automated Validation and Centralized Data: The Future of CPV Compliance
Shift CPV from manual reports to real-time control with automated validation and centralized data. ...
Read
Smarter by the Second: How Real-Time Monitoring is Redefining Pharma and Biopharma Manufacturing
Catch process drift early with real-time monitoring, support PAT, and improve pharma manufacturing ...
Read
The Path to Digital CPV
Start digital CPV with real-time trending, alarms, and compliant records. Cut manual effort and ...
ReadWebinars

How ValGenesis Process Lifecycle Suite Frees Up Teams to Focus on Quality, Not Data Entry
Continued Process Verification (CPV) was introduced with the FDA’s 2011 Process Validation guidance ...
Watch
Digital CPV: Important Step in Your Pharma 4.0 Journey
Digitization is an objective for organizations that want to be compliant, agile, sustainable and ...
Watch
Digital Transformation of Your Site: A Step-By-Step Program
Digitalization of companies operating within the life-science landscape has gained strong momentum ...
Watch
Avoid CPV Failures: Proven Ways to Catch Deviations Early
In its 2011 Guidance for Industry: Process Validation—General Principles and Practices, the FDA ...
Watch
Reducing Errors and Gaining Efficiency in Your CPV Program
The concept of continued process verification (CPV) was first introduced a decade ago, when the FDA ...
WatchVideos
ValGenesis iCPV™: Proactive Monitoring. Real-Time Insights. Quality Assured.
See how real-time CPV monitoring flags CPP/CQA trends early and automates reports. Watch the iCPV ...
Watch
Shift from Reactive to Proactive: A 2-Minute Intro to Smarter CPV
Move CPV from reactive to proactive with real-time visibility, automated workflows, and consistent ...
Watch
Manual CPV vs Digital CPV: Why Should you Upgrade?
Compare manual vs digital CPV and see how real-time, predictive monitoring improves quality control ...
Watch
Spend Time on Quality, Not Paperwork: Gaining Efficiency with Digital CPV
See how digital CPV automates monitoring, detects deviations early, and frees SMEs from manual ...
WatchFrequently Asked Questions
Ready to Automate
Real-time Process Monitoring?
Book a demo to discover how a fully digital CPV can impact your processes, your manufacturing, and your product quality.