Process Excellence.
From Design to Verification.
ValGenesis Process Lifecycle Suite combines iCMC and iCPV to unify the process lifecycle - powering smarter decisions, seamless tech transfer, and sustained product quality.

The Upgrade CMC Deserves.
Join the life sciences leaders using the ValGenesis Process Lifecycle Suite to remove barriers to innovation and accelerate delivery - by making process lifecycle execution consistent, connected, and efficient.



Customer Testimonials

We work with ValGenesis iCMC to digitalize our QbD framework within GSK for the development of new vaccines. We are confident that iCMC will be key for simplification, acceleration, and knowledge management.
Hervé Gressard
Head of Global Project Statistics CMC Technical R&D

Implementing ValGenesis iCPV has transformed our approach to CPV. The integration of our data systems and the comprehensive training from the ValGenesis team have empowered our users, ensuring we meet regulatory standards while enhancing efficiency and data integrity.
Patricia Watanabe
Validation Supervisor
Used By Life Sciences Leaders Worldwide
Process
Lifecycle Suite
Process Excellence.
From Design to Verification.

iCMC
Design robustness, not rework, with ValGenesis iCMC™. Centralized QbD tools link QTPP targets to CQA/CPP analysis, FMEA and HAZOP risk scoring, and ML-powered trend analytics in one traceable hub. Harmonize processes across products, satisfy regulators with confidence, accelerate development, and get to market first.
Learn More
iCPV
Join the world of real-time monitoring with ValGenesis iCPV™. Multivariate analytics, SPC control charts, and guided workflows transform siloed data into early-warning insights, automating CPV reports across sites and batches. Detect drifts before they hurt quality, maintain continuous readiness, and future-proof production - explore iCPV now.
Learn MoreScience and data-driven process design
Apply QbD and risk-based frameworks to build quality into every stage of drug development.Real-time performance oversight
Monitor CPPs and CQAs continuously to detect trends and ensure consistent product quality.Confidence in change control
Support post-approval changes with data-driven risk assessment and full traceability.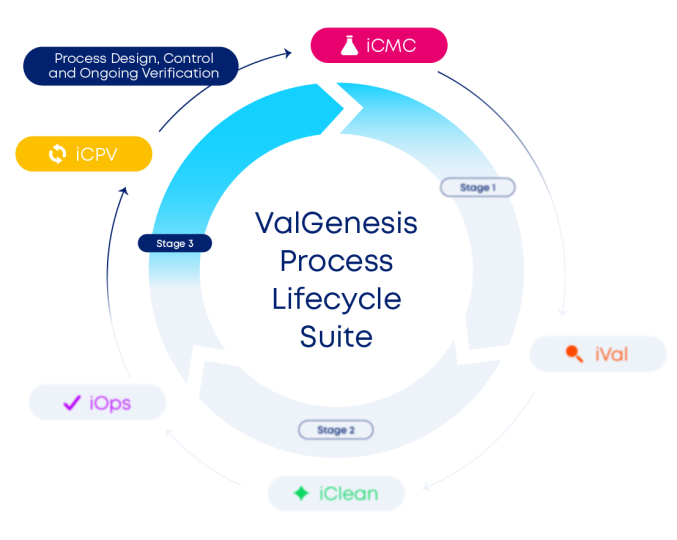
Resources

Integrating Digital Transformation and Quality by Design for Enhanced Pharmaceutical Development
Discover how digital platforms paired with QbD speed pharma development and support ICH compliance. ...
Read
Digital Transformation in CDMOs: Streamlining Tech Transfer Processes
Reduce tech transfer delays in CDMOs with digital workflows and data transparency. Get the pitfalls ...
Read
Risk and Data as Knowledge Enablers: A Lifecycle Approach
Turn pharma lifecycle data into usable knowledge with KASA-aligned structure and risk workflows. ...
Read
Avoiding the Pitfalls of PQR
Avoid APQR pitfalls with a digital CPV approach that supports real-time reviews. Read this Industry ...
ReadWebinars

Introducing ValGenesis Process Lifecycle Suite
Life sciences organizations are shifting from siloed systems to unified platforms, not only to meet ...
Watch
Revolutionizing CMC in Drug Development: How to Reduce Errors and Delays in Quality by Design
Quality by Design in pharma offers significant potential—but many organizations struggle to ...
Watch
Digital Transformation of Your Site: A Step-By-Step Program
Digitalization of companies operating within the life-science landscape has gained strong momentum ...
Watch
AI and Machine Learning as Enablers for Industry 4.0
While AI adoption offers significant potential, the industry is still transitioning from ...
WatchVideos
ValGenesis PLS™: Process Excellence. From Design to Verification.
Modernize process development and CPV with QbD workflows, real-time monitoring, and risk templates. ...
Watch
ValGenesis iCPV™: Proactive Monitoring. Real-Time Insights. Quality Assured.
See how real-time CPV monitoring flags CPP/CQA trends early and automates reports. Watch the iCPV ...
Watch
ValGenesis iCMC™: Science-Driven. Risk-Controlled. Designed for Robustness.
Replace scattered CMC docs with QbD-based, risk-controlled workflows in ValGenesis iCMC—watch now ...
Watch
Don’t Let Spreadsheets Hold Back Your CMC: How ValGenesis Process Lifecycle Suite Transforms QbD Efficiency
Move CMC development beyond spreadsheets. See how a digital QbD platform structures risk, connects ...
WatchFrequently Asked Questions
Do you need help or have questions about any features?
End-to-End Process Excellence
Book a demo today to enable smarter decisions, streamline tech transfers, and assure consistent product quality.
Implement science-based design, real-time oversight, and confident change management—all in one unified platform.