AI-Powered Validation. From Planning to Execution.
ValGenesis Validation Lifecycle Suite connects iVal, iClean, and iOps in a single digital platform—standardizing validation, accelerating time to market, and ensuring audit readiness across the enterprise.

Global Adoption. Powerful Results.
Join the industry leaders who are replacing disconnected documents and silos with a unified validation lifecycle approach.













Customer Testimonials

We chose ValGenesis because they were a known and trusted vendor. We had limited time and resources, and we knew the quicker we could implement the solution, the sooner we’d start realizing value from it.
Joshua Pelina
Validation Engineer

Working with ValGenesis was great. They helped us align the system with our new CSV program and uncover opportunities for process improvements and efficiency gains to maximize value.
Jeff Onis
Global IT Data Integrity Manager
Used By Life Sciences Leaders Worldwide
Validation
Lifecycle Suite
AI-Powered Validation.
From Planning to Execution.

iVal
Supercharge validation with ValGenesis iVal™. AI-powered authoring, automated execution, live anomaly flags, and bulletproof traceability slash cycles by up to 80% and cut observations by 90%. From CQV to CSA, you’ll be audit-ready and market-ready faster than ever. iVal takes digital validation to the next level.
Learn More
iClean
Halve cleaning validation timelines and wipe out manual math with ValGenesis iClean™. Automated MACO calculations, ADE-aligned limits, 2D/3D equipment maps, and rule-driven workflows deliver digital, inspection-ready files across all sites. Standardize globally, flex locally, and gain real-time oversight - discover iClean today.
Learn More
iOps
Leave paper logbooks behind with ValGenesis iOps™. Mobile, QR-enabled forms capture every use, cleaning, and calibration event in real time; automated reviews, deviations, and alerts seal compliance gaps. Achieve 100% traceability, trim documentation labor by 70%, and connect ops to quality with iOps today.
Learn MoreUnified, End-to-End Validation
Manage the entire CQV, CSV, and CSA lifecycle in one centralized, standardized suite.Continuous Compliance and Traceability
Ensure audit readiness with secure, real-time validation records, impact assessments, and change management oversight.Smarter, Paperless Workflows
Replace manual tasks and siloed tools with intelligent, automated processes that ensure teams are always ready to execute.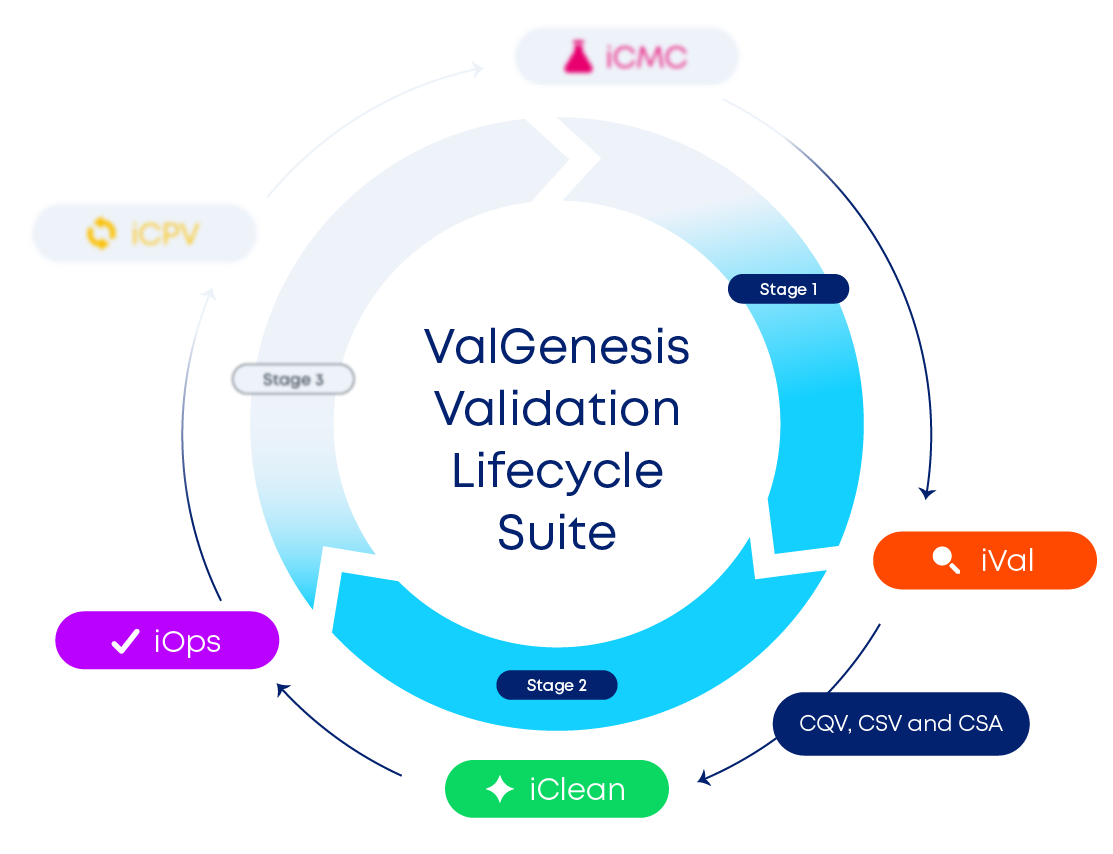
Resources

Ensuring Trust in AI: The ValGenesis Approach to Validating AI Agents and Functions for Validation and Qualification in Life Sciences
Validate AI Smart Agents for GxP work with a practical, compliance-first framework. Read the white ...
Read
Top 10 Fastest Benefits of Paperless Validation
Cut validation time, reduce storage costs, and speed audits with paperless validation. Read the top ...
Read
The Playbook for Truly Paperless Validation in Pharma
Cut validation time and errors with digital workflows. Learn the steps to go paperless and be ...
ReadWebinars

Introducing ValGenesis Validation Lifecycle Suite
ValGenesis Validation Lifecycle Suite (VLS) is a step-change in life sciences validation software. ...
Watch
Meet VAL and Take Advantage of AI-Driven Validation
Register for this webinar to see how VAL, the AI-driven Validation Assistant, can transform the way ...
Watch
AI Powered Validation: How to be Smarter, Faster, and More Reliable
Traditional validation processes can slow teams down with manual reviews, paper-driven workflows, ...
Watch
Why is Manual Validation Holding You Back? The Evolution of AI-Driven Validation in Regulated Industries
Manual validation has long been the industry standard, but it often slows teams down, introduces ...
WatchVideos
ValGenesis VLS™: AI-Powered Validation. From Planning to Execution.
Digitize qualification, cleaning validation, and operations with an AI-assisted suite—watch the ...
Watch
Types of Validation in the Pharmaceutical Industry
Learn the main types of pharmaceutical validation across development and manufacturing and ...
WatchFrequently Asked Questions
ValGenesis VLS features advanced impact assessment functionality, specifically designed to streamline change management throughout the lifecycle of a system, equipment, or process. When a change is required, users initiate the impact assessment process by selecting the necessary updates, after which the system automatically identifies the directly and indirectly impacted specifications and tests using the dynamic requirements trace matrix.
Beyond impact determination, VLS enables modifications exclusively to affected requirements, specifications, and tests, eliminating the need for individual document revisions. Additionally, the system possesses a unique capability to generate a consolidated document from impacted components, significantly reducing the time required to manage changes of any scale.
Ready to Accelerate Your Validation Lifecycle?
Book a demo today and unify your validation lifecycle in one AI-powered platform. Standardize workflows, ensure continuous compliance, and accelerate time to market.