
ValConnect Innovation Day Princeton: A Glimpse into the Digital Future of Life Sciences
ValConnect Innovation Day Princeton brought Bayer, GSK, Cardinal Health, Transcat & Hansa Biopharma together to explore AI & Smart GxP™ in life sciences.
Welcome to the ValGenesis blog, your resource for timely insights from industry-leading experts on the trends, best practices, and new technologies impacting the life sciences.
Nov 30, 2023 2:12:33 PM | Explore the key aspects of ICH Q12 and a 5-step structured framework for effective post-approval change management (PACM) agility.

Explore the key aspects of ICH Q12 and a 5-step structured framework for effective post-approval change management (PACM) agility.

Learn how to simplify regulatory approval for post-approval changes and discover the benefits of implementing a PACM protocol and using a QRM platform.

Today we want to take a fresh look into comparability for post-approval changes. How? We're thinking of a unique multiparametric approach.

In this blog post, we will give you all the steps about how to create a PACM protocol and how ICH Q12 supports you in it.

Quality Risk Management
Finding the Root Cause in mAB Production: a ValGenesis Story
Learn how we helped a customer find the root cause of a problem in monoclonal antibody (mAB) production and become stronger.

Written by
Sofia Santos

Tech Transfer & Process Scale-up
How to Accelerate Process Scale-Up With Effective Tech Transfer
There's a need to accelerate pharma product development and process scale-up. Having an effective tech transfer strategy can help you.

Written by
Sofia Santos
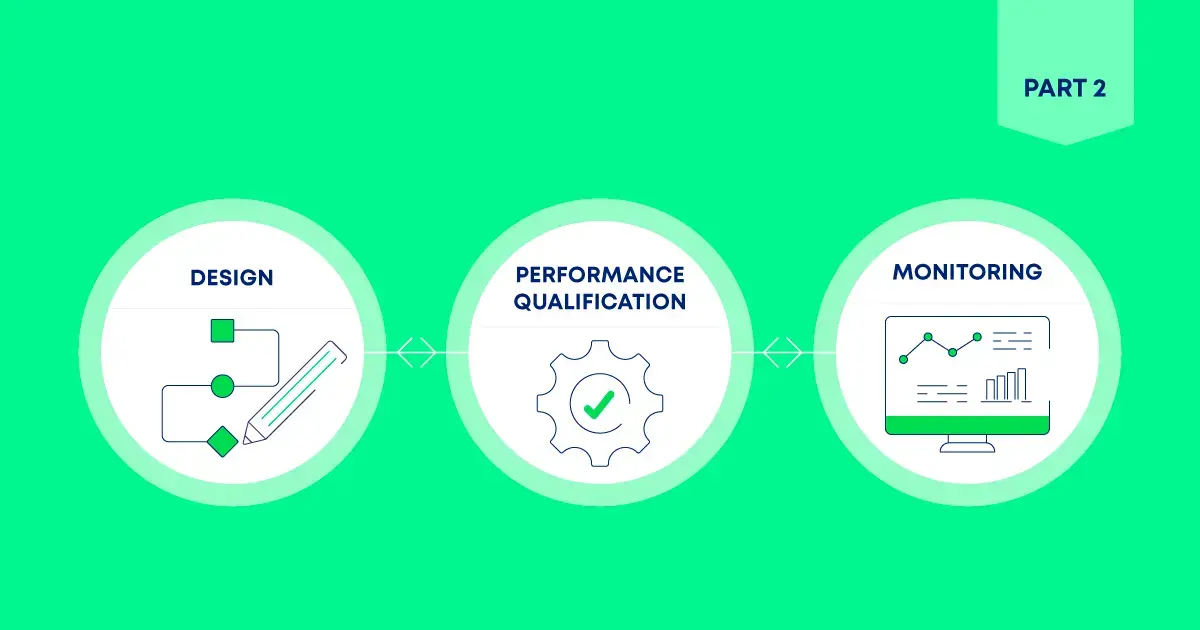
Cleaning Validation
Risk-Based Cleaning Validation: A Lifecycle Approach
Explore the evolution of pharmaceutical cleaning validation from compliance-focused practices to a risk-based, lifecycle-driven approach.
Written by
Kenneth Pierce
CSA flips the paradigm of traditional CSV to "right-size" validation processes. Learn more about the benefits and principles of the CSA approach, which encourages critical thinking and automation.

ValConnect Innovation Day Princeton brought Bayer, GSK, Cardinal Health, Transcat & Hansa Biopharma together to explore AI & Smart GxP™ in life sciences.
Previously
Catalent's Journey of Continuous Improvement for CSV Excellence
4 Conversations to Drive Your Business Case for Digital Validation
Learn how digitization offers a consistent way to carry out cleaning validation lifecycle activities, ensuring your methods and procedures are risk-based and rooted in scientific evidence.
.webp)
Discover how ValGenesis iClean™ standardizes change impact assessments in cleaning validation, enhancing consistency and compliance across teams and sites.
Previously
Unlocking Operational Capacity with Fully Digitalized Cleaning Validation
The Real Cost of Bottlenecks: Why Manual Cleaning Validation Increases Regulatory Risks
Waves of new technology and thinking — IoT, AR, cloud, Agile — are transforming the very foundation of validation processes. Learn how to leverage them in a Pharma 4.0 world.
Join our newsletter to receive updates on the latest news and industry-related content tailored to your preferences.
