ICH Q12 Implementation: Enhancing PACM Agility


A Structured Framework for Successful ICH Q12 Adoption
In the ever-evolving landscape of pharmaceutical manufacturing, the implementation of ICH Q12 presents a significant opportunity for companies to enhance their post-approval change management (PACM) and improve their agility in providing quality medicines to patients. While several companies have embarked on pilots to embrace this guideline, many have yet to fully realize the benefits it can offer. In this blog post, we will explore the key aspects of ICH Q12 and how companies can proactively adopt its principles.
Introduction to ICH Q12
Published in 2019, ICH Q12 aims to provide a structured approach to increase flexibility for post-approval changes in commercial products. It builds upon the foundation laid by its predecessors, including ICH Q8, Q9, Q10, and Q11, promoting continuous improvement, innovation, and accelerated drug product supply. By employing a science- and risk-based approach, ICH Q12 empowers companies to navigate changes in the product lifecycle effectively.
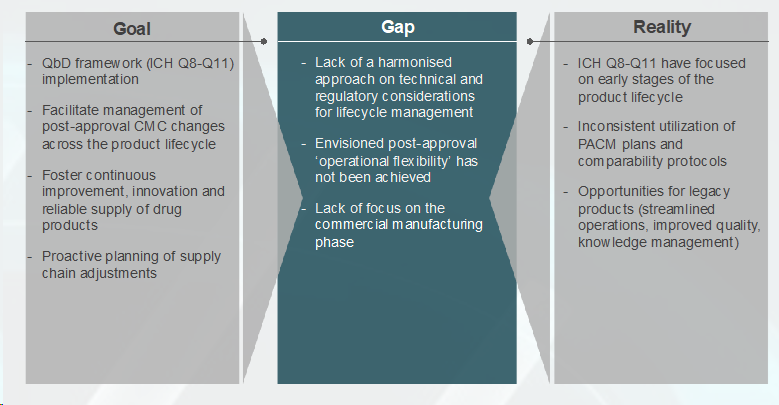
Figure 1 — Schematic of how ICH Q12 bridges an industry vision with the current reality.
Change is a catalyst for progress, leading to improved product quality and innovation and enhanced product economics. As companies manage changes, they also cultivate a culture of quality and continuous improvement, from the early stages of product development to discontinuation. Anticipating predictable changes, such as capacity expansion or technology advancements, is an integral part of the process.
Digitalization: Streamlining ICH Q12 Implementation
Digital platforms play a vital role in the seamless application of ICH Q12. They offer a standardized approach to risk assessment and data analysis, ensuring data integrity and regulatory compliance and fostering knowledge sharing and collaboration. This digital transformation can help companies extend the benefits of ICH Q12 to a wide range of products within their organization.
Why was ICH Q12 Created?
ICH Q12 bridges the gap between the pharmaceutical industry's vision of efficient changes in commercial products and the current regulatory environment, which is often marked by excessive scrutiny and formality. The guideline shifts the paradigm from "Tell and Do" to "Do and Tell." As of now, companies have to seek regulatory approval before implementing post-approval changes, leading to a "Tell and Do" approach. ICH Q12 introduces a more agile "Do and Tell" approach, where certain well-defined changes can be implemented without extensive regulatory submissions, with notifications following the change.
The Foundational Requirements for ICH Q12
ICH Q12 integrates key concepts from other ICH guidelines. To apply it effectively, companies should embrace the principles and requirements of these guidelines early in the product lifecycle.
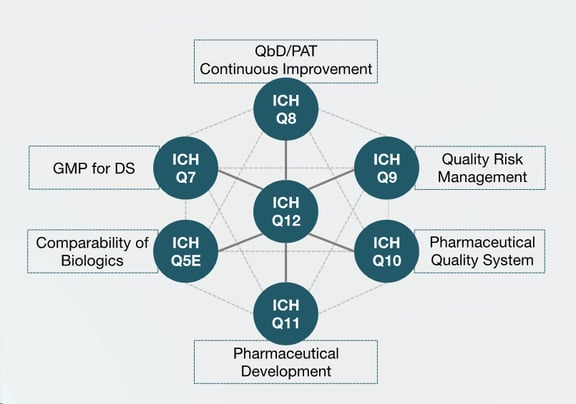
Figure 2 — Overview of the relationship of ICH Q12 to other ICH guidelines.
For example, the importance of following a QbD approach (ICH Q8) and QRM (ICH Q9) across all stages of process development, including the definition of design space and the impact of the process inputs on the critical quality attributes (CQAs), will greatly simplify past approval change as it facilitates a better understanding of the impact of the change on the product quality/process performance. The body of process and product knowledge is built up during the development and commercial stages.
For legacy commercial products, a retrospective approach can be considered, which will enable the application of the ICH Q12 principles.
The Framework for Post-Approval Change Management Agility
A structured framework is essential for effective PACM agility. It includes five main steps:- Criticality Analysis: Assess the critical aspects of the current process, including critical process parameters (CPPs), CQAs, and control strategy.
- Risk Impact Assessment and Prioritization: Identify and prioritize risks, considering their impact on the process and product quality.
- Gap Analysis: Determine areas where further studies are needed to understand the impact of proposed changes.
- Control Strategy Revision: Update the control strategy based on the risk assessment and gap analysis.
- Continued Process Verification and Lifecycle Management: Utilize the PACM protocol to manage established conditions (EC) and regulatory communication.
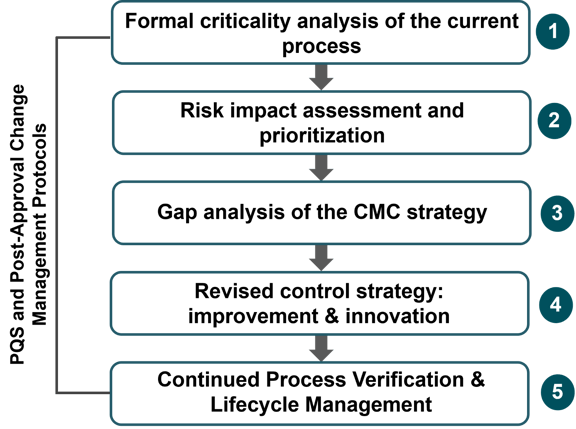
Figure 3 — A structured end-to-end framework for lifecycle management of legacy products. Post-approval changes are part of product lifecycle management. Adapted from Strohmeier et al., 2018.
Challenges of Implementing ICH Q12
Implementation of ICH Q12 poses certain challenges, such as the need for thorough risk assessments, personnel training, and data collection, as well as the need to address regional regulatory variations. Digitalization can alleviate some challenges, streamlining risk assessment, data analysis, and regulatory compliance.
Conclusion
ICH Q12 offers a harmonized, flexible approach to PACM, empowering companies to adjust products and processes and supply medicines more efficiently. By proactively managing changes, companies can enhance product quality, contribute to patient safety, and gain a competitive edge. The structured framework outlined in this post can help companies successfully implement ICH Q12, with digitalization further supporting their efforts.
The path to successful ICH Q12 adoption begins with embracing change and implementing a robust framework that combines science, risk-based approaches, and digital solutions. This journey promises to lead to improved pharmaceutical processes and better patient outcomes.
Related Blog Posts

AQbD FAQs: Practical Answers to ATP, MODR, ICH Q14, and more
Get direct answers to common AQbD questions, from ATP and MODR to analytical control strategy, lifecycle management, and practical ICH Q14 implementation.
By Sofia Santos

Creating a Post-approval Change Management (PACM) Protocol
In this blog post, we will give you all the steps about how to create a PACM protocol and how ICH Q12 supports you in it.
By Ricardo Leandro
Why QbD Breaks Down After Approval (and What Leaders Miss)
Learn why Quality by Design (QbD) often fails post-approval and how pharma leaders can ensure decision traceability for continuous improvement and success.
By Sofia Santos