
ValConnect Innovation Day Princeton: A Glimpse into the Digital Future of Life Sciences
ValConnect Innovation Day Princeton brought Bayer, GSK, Cardinal Health, Transcat & Hansa Biopharma together to explore AI & Smart GxP™ in life sciences.
Welcome to the ValGenesis blog, your resource for timely insights from industry-leading experts on the trends, best practices, and new technologies impacting the life sciences.
Aug 17, 2023 12:40:33 PM | Discover how machine learning unlocks drug developability predictions' potential in a successful case study.

Discover how machine learning unlocks drug developability predictions' potential in a successful case study.

Unlocking the potential of Computer System Assurance (CSA) in pharmaceuticals: A roadmap for a seamless transition from CSV to CSA.

Blockchain technology is revolutionizing how data integrity is ensured in clinical trials, establishing a new level of trust and compliance.
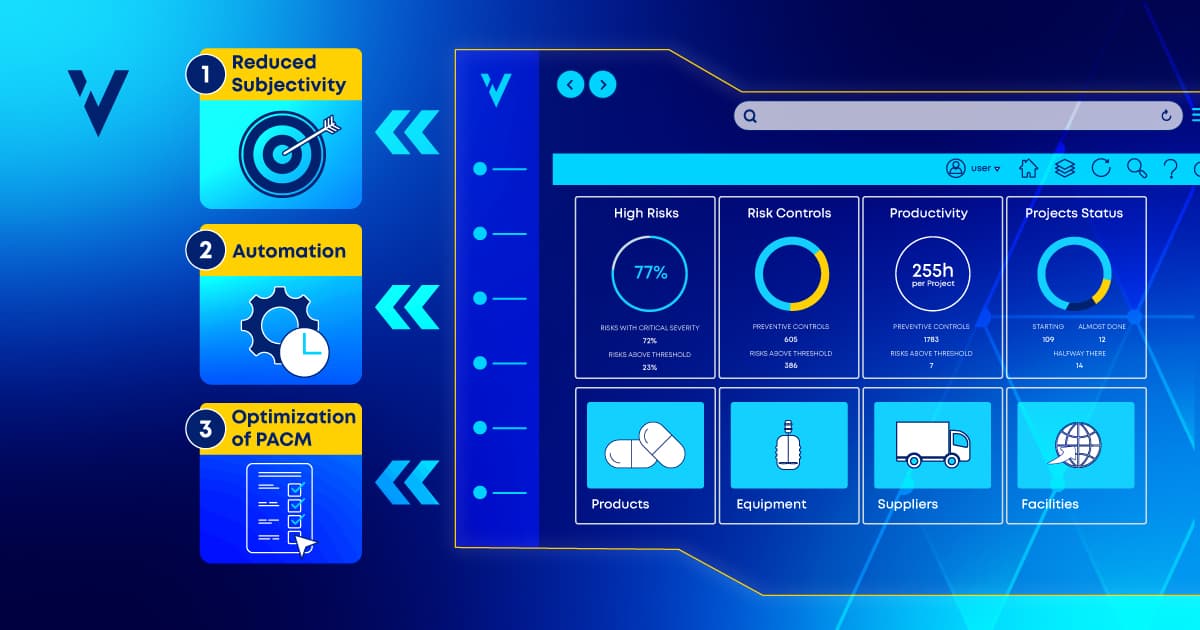
In this post, we’ll look at 3 benefits of integrating process data with risk management and how it increases organizational efficiency and competitiveness.

How do we deliver objective evidence with a high degree of certainty it will withstand regulatory scrutiny? Technology tools like a VLMS hold the answer.

Learn about the requirements of the Annex 1 revision and how to ensure compliance with a digital risk management platform.

In this blog post, we will explore how making your transition from the traditional CSV to the new CSA approach can save you time and resources.

Sponsors and CROs are prioritizing digitization to build a foundation for decentralized clinical trials and using AI/ML tools to improve patient outcomes.

In this blog post we'll look into what steps you may need to take to make sure you are compliant with the ICH Q9(R1) guideline.

Learn how implementing digital CPV plans can contribute to continuous processes reaping the benefits of Industry 4.0 and digitalization.

Spreadsheet tools like Excel raise a regulatory red flag and can trigger an audit. Your best defense is a validation lifecycle management system (VLMS).

ValGenesis Process Manager can help life sciences companies avoid FDA 483s and establish a harmonized cleaning validation program companywide.

Explore the three types of quality risk management (QRM) tools in pharma and learn how to assess, prioritize and control risk across the product lifecycle.
.webp)
Learn how to write clear, defensible requirements and use Agile methodology and structured qualifiers to strengthen traceability in CSV and CSA.

In this blog post, we want to discuss about the application of the Quality by Design for Cell & Gene Therapies. Are they suited for them?

Quality Risk Management
Finding the Root Cause in mAB Production: a ValGenesis Story
Learn how we helped a customer find the root cause of a problem in monoclonal antibody (mAB) production and become stronger.

Written by
Sofia Santos

Tech Transfer & Process Scale-up
How to Accelerate Process Scale-Up With Effective Tech Transfer
There's a need to accelerate pharma product development and process scale-up. Having an effective tech transfer strategy can help you.

Written by
Sofia Santos
Cleaning Validation
Risk-Based Cleaning Validation: A Lifecycle Approach
Explore the evolution of pharmaceutical cleaning validation from compliance-focused practices to a risk-based, lifecycle-driven approach.
Written by
Kenneth Pierce
CSA flips the paradigm of traditional CSV to "right-size" validation processes. Learn more about the benefits and principles of the CSA approach, which encourages critical thinking and automation.

ValConnect Innovation Day Princeton brought Bayer, GSK, Cardinal Health, Transcat & Hansa Biopharma together to explore AI & Smart GxP™ in life sciences.
Previously
Catalent's Journey of Continuous Improvement for CSV Excellence
Are You Aligned with FDA's Computer Software Assurance Methodology?
Learn how digitization offers a consistent way to carry out cleaning validation lifecycle activities, ensuring your methods and procedures are risk-based and rooted in scientific evidence.
Explore the complexities of cleaning validation limits, including HBEL/MACO, 10 ppm, and visually clean to ensure patient safety and product quality.
Previously
Change Impact Assessments in ValGenesis iClean™: A Framework for Consistency
Unlocking Operational Capacity with Fully Digitalized Cleaning Validation
Waves of new technology and thinking — IoT, AR, cloud, Agile — are transforming the very foundation of validation processes. Learn how to leverage them in a Pharma 4.0 world.
Join our newsletter to receive updates on the latest news and industry-related content tailored to your preferences.
