
ValConnect Innovation Day Princeton: A Glimpse into the Digital Future of Life Sciences
ValConnect Innovation Day Princeton brought Bayer, GSK, Cardinal Health, Transcat & Hansa Biopharma together to explore AI & Smart GxP™ in life sciences.
Welcome to the ValGenesis blog, your resource for timely insights from industry-leading experts on the trends, best practices, and new technologies impacting the life sciences.
Apr 20, 2023 9:52:52 AM | ValGenesis (VLMS) has capitalized on delivering functionality to support object-oriented validation. Here you can create requirements as objects, and these can be shared.

ValGenesis (VLMS) has capitalized on delivering functionality to support object-oriented validation. Here you can create requirements as objects, and these can be shared.

This post is about tips to write your User Requirements Specification and how it can improve your processed data and software.
-1.jpg)
Pharma has embraced digitization in many business lines, but validation has been one of the few processes to remain manual. Here are 12 reasons to make the switch! (Part 1 of 2)

Explore the top 10 business benefits you'll receive from digitizing your paper-based validation processes with ValGenesis.

A better resource use, a simpler process, and a superior product quality. These are some of the CSA benefits explored in this blog post.

Cloud-based digital validation allows you to recover in the event of a disaster, ensuring business continuity and compliance.

In this blog post, we show you how digital validation can improve your validation process and enhance your business efficiency.

This post examines four reasons tablet computers are ideal for validating computer systems, equipment, and instruments in controlled GMP manufacturing environments.

Faster innovation and time to market are top priorities for CDMOs. Being a digital-first organization gives Theragent a substantial competitive advantage.

ROI study reveals how much time and money customers save by digitizing corporate validation processes. Watch the webinar (link in post).

Saving bottom-line costs of paper and paper storage is just one benefit of paperless validation. Also at stake: disaster recovery, using big data intelligently, and keeping remote teams productive.

Root-Cause Analysis
Finding the Root Cause in mAB Production: a ValGenesis Story
Learn how we helped a customer find the root cause of a problem in monoclonal antibody (mAB) production and become stronger.

Written by
Sofia Santos

Tech Transfer & Process Scale-up
How to Accelerate Process Scale-Up With Effective Tech Transfer
There's a need to accelerate pharma product development and process scale-up. Having an effective tech transfer strategy can help you.

Written by
Sofia Santos
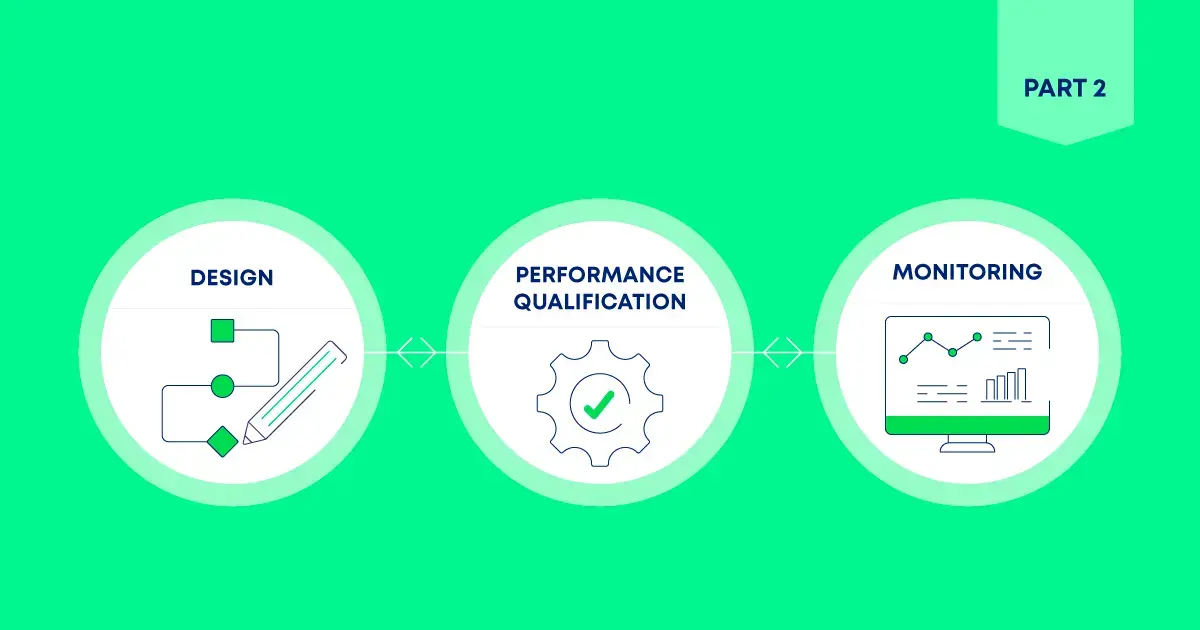
Cleaning Validation
Risk-Based Cleaning Validation: A Lifecycle Approach
Explore the evolution of pharmaceutical cleaning validation from compliance-focused practices to a risk-based, lifecycle-driven approach.
Written by
Kenneth Pierce
CSA flips the paradigm of traditional CSV to "right-size" validation processes. Learn more about the benefits and principles of the CSA approach, which encourages critical thinking and automation.

ValConnect Innovation Day Princeton brought Bayer, GSK, Cardinal Health, Transcat & Hansa Biopharma together to explore AI & Smart GxP™ in life sciences.
Previously
The Benefits of Automating Your Requirements Traceability Matrix
Catalent's Journey of Continuous Improvement for CSV Excellence
Learn how digitization offers a consistent way to carry out cleaning validation lifecycle activities, ensuring your methods and procedures are risk-based and rooted in scientific evidence.

Discover how fully digitalized cleaning validation boosts efficiency, ensures compliance, and transforms operational capacity in pharmaceutical manufacturing.
Previously
The Real Cost of Bottlenecks: Why Manual Cleaning Validation Increases Regulatory Risks
Digital Cleaning Validation: From Lifecycle Control to Real-Time Assurance
Waves of new technology and thinking — IoT, AR, cloud, Agile — are transforming the very foundation of validation processes. Learn how to leverage them in a Pharma 4.0 world.
Join our newsletter to receive updates on the latest news and industry-related content tailored to your preferences.
