Top 10 Fastest Benefits of Paperless Validation

Summary
This e-book explains why paper-based validation still creates wasted time, audit friction, and frustration in life sciences, and why modern electronic validation is different from early, costly e-validation tools.
It outlines 10 practical benefits of going paperless, including reduced paper/storage burden, faster protocol work, automated change control, easier collaboration, and quicker inspections and audits.
Key takeaways
- Paperless validation reduces paper, shredding, and storage demands and can free up facility space for labs and equipment.
- Electronic workflows speed authoring, execution, reviews, approvals, deviation handling, and record retrieval through centralized, searchable records.
- Automation improves change control, revision tracking, collaboration, audit readiness, and scalability across sites.
Who is this for
- Validation engineers / CQV professionals (CSV, equipment, process, cleaning validation)
- Quality assurance (QA) managers and document control leads
- Manufacturing engineering and technical operations leaders in regulated facilities
- Regulatory compliance and audit/inspection readiness teams
- Laboratory managers and QC/analytical operations leaders
- IT/Systems owners supporting regulated platforms and electronic records
- Project/program managers overseeing validation timelines and cross-site work
Download your e-Book
Top 10 Fastest Benefits of Paperless Validation
We are living in a paperless world.
Our day-to-day electronic activities, from banking apps to text messaging to maintaining an appointment calendar, have eliminated the need for paper records.
But many businesses hold onto paper. For example, validation processes in many life sciences organizations remains a paper-centric process. This creates unnecessary audit pain, wasted time and worker frustration. Often, the refusal to go paperless stems from lingering misconceptions about why a paperless validation solution would be necessary, worthwhile or even beneficial.
In the early days after FDA 21 CFR Part II went into effect, many life sciences organizations refused to adopt electronic validation. In those days, there were valid reasons for this decision. The cost of implementing regulation-compliant e-validation solutions (those that were even available) was significantly higher than simply retaining paper-based validations.
Specialized, purpose-built platforms did not yet exist. What solutions that did exist did little more than store executable PDFs on CDs. Appropriately, many life science organizations saw electronic validation as an extremely expensive that offered little to no tangible return. A lot has changed since then.
Electronic validation lifecycle management systems have evolved into purpose-built powerhouses of functionality. The old arguments no longer hold. The benefits of “paperless” go beyond just paper—it’s about digitization, collaboration, analysis, scalability and total business performance.
1. Less paper waste and less storage costs
Paper isn’t free. Not even close. For life sciences companies, these costs add up.
More importantly, the paper that a company does use mostly goes to waste. According to IVT Network, even if a typical life sciences company purchases 10 million sheets of paper annually, 95% of this paper will be disposed of, and the rest will ends up in filing cabinets1. It’s not just the cost of purchasing paper but also the costs of secure shredding, disposal and storage.
These costs are greatly reduced for those who adopt a paperless validation solution. In fact, according to a WRAP study on business efficiency2, a paperless solution can reduce these costs by approximately 65% even if some paper is still allowed.
If you are maintaining paper records, you’re going to house them in a secure physical space. This means filing cabinets— a lot of filing cabinets. The storage demands can quickly become unsustainable when the average worker is generating more than 45 pages per day3.
Without rooms of filing cabinets, your company can build additional labs, house more testing equipment, or increase storage for test samples. Every square foot a facility must dedicate to the storage of paper records is one less dedicated to actual validation activities.
1) King-Bailey, Valerie. “Mastering Regulatory Compliance”
2) WRAP. Green Office
3) “Paperless document management saves time, money and the environment”
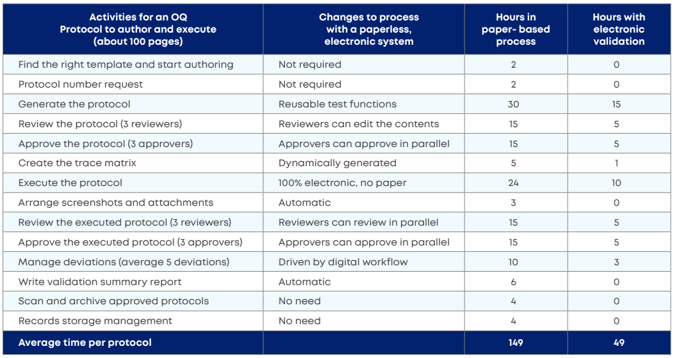
2. Triple-time productivity
No one disputes that paper-based processes, no matter how optimized, are tedious, lengthy and linear. The advantages of an electronic solution are clear: digital workflows, electronic signatures, task notifications, and more.
Here is how time-saving efficiency materializes in a typical protocol.
3. Smarter change control and revision tracking
For life sciences companies using traditional paper-based validation systems, change control and document revision tracking are extremely time-consuming tasks.
This is because paper-based systems aren’t automated. Before a change can be implemented, a change control request form must be physically printed and filled out. It must then be submitted to a change control committee. That committee must then set up a meeting to review/discuss the proposed change and decide whether to approve or reject the request. Only after committee approval can the change be implemented.
If the person deviates from the change control plan submitted in the request form, they will have no choice but to submit another change request form and start all over again.
In paper-based processes, access to records is restricted to the number of physical copies. Records must be signed out/ in with a logbook, which must also be managed. This means hiring an administration/records manager, which is yet another operating cost. If someone needs to view a record that has already been signed out, and no more copies are available, they must then waste even more time physically tracking down the person who last signed it out. Even then, the record cannot change hands until the logbook has been properly updated.
This is terribly convoluted and frustratingly time-consuming. It’s also completely unnecessary.
A modern electronic validation lifecycle management system could easily automate the change control and document tracking processes through instant digital workflows and electronic approvals. With a “closed-loop” change management approach, change impacts can be instantly evaluated across the entire validation lifecycle.
4. Improved collaboration and communication
In any life sciences validation environment, it’s unlikely (maybe impossible) that you could find a single person with the knowledge and skills necessary to execute a validation process independently without help from colleagues.
In any lab, testing or manufacturing facility, teams work collaboratively. For optimal productivity, you need clear communication facilitated by user-friendly collaboration tools.
Collaboration means versions and revisions constantly, physically changing hands between team members in a paper-based system. It’s difficult to track status. In-person meetings, phone calls and emails are wasted trying to get on the same page.
A paperless validation solution eliminates these headaches. And it doesn’t matter whether colleagues are sitting next to one another or collaborating from different sites or even home offices. Electronic tools expedite communication, and automated routing simplifies revisions and change tracking.
For example, workflow automation fires off task notifications, schedules and nudging features through email, and simply clicking on those links can keep the process moving. And with information centralized and digitized, real-time collaboration— combined with real-time analysis of the process and automated trace matrix generation—ensures records are complete and compliant.
5. Smarter task and team management
Even in a paper-based validation system, there are still some electronic elements. This is especially true in task and team management.
However, these tools are limited—usually done via company email instead of in a centralized hub or dashboard. This makes management far less efficient.
However, with a paperless/electronic solution, team members and tasks can be easily managed and tracked because everyone is assigned and performs their assigned tasks on the same centralized platform. As a result, team leaders can assign, view and update tasks all from the same dashboard, giving a comprehensive overall view of progress to optimize team performance.
And the electronic workflow that routes tasks and information between team members can be easily modified to accommodate a change in process or set up with documented deviation rules. All within the framework of a FDA 21 CFR Part 11-compliant feature set that includes time-stamped audit trails, reporting and electronic signature capabilities.
6. Recyclable and reusable content
Reuse of content is a great timesaver. In a paper-based system, the reusability of content is often restricted to templates and standard forms.
However, past documents cannot be used to create new ones. In a traditional paper-based system, validation documents tend to be rewritten ‘from scratch’ each time.
A paperless validation solution can easily reuse previous content such as requirements, test cases, and more which reduces the time and effort involved in producing validation documentation.
Paperless validation solutions also have templates and forms. However, they also commonly offer tools that give users the ability to access, reuse/recycle, and manipulate pre-existing documents to create new or edited content. As a result, new documents can often be created faster and with greater levels of consistency.
7. Streamlined review and approval process
For paper-based validation processes, reviews and approvals require physically tracked transfers of revised documentation.
Documents must be physically signed in and out; email notifications must be manually written and sent by personnel; revision tracking requires interpersonal communication and manual verification, and status tracking is dependent on the project management skills of team leaders and management.
The sorts of “line through” edits that are standard with paper cannot accurately display precisely when and by whom specific changes were made. These pain points translate into many wasted work hours which leads to unnecessarily excessive validation timelines.
An electronic, paperless validation solution offers organizations many useful tools that easily and conveniently eliminate paper-based pain points. Revisions, once made, are automatically routed to reviewers and approvers. Email notifications and task alerts are automatically sent to appropriately assigned personnel. Statuses are automatically updated, and document/task progress is tracked via user dashboards. Wasted work hours that would be found in a paper-based system are fully minimized with a paperless solution as the review/approval process is streamlined and far more efficient.
8. Faster search and retrieval
Unless you know the exact document you need (and, in some cases, its precise title), the search and retrieval process is difficult and time-consuming in a paper-based validation system.
While topic-based and project-based filing standards can ease this burden somewhat, paper-based methods are still painfully slow in comparison to the document search/retrieval tools that are usually available in an electronic validation solution. And what organizations frequently discount is the effort and work hours necessary to create and maintain a competent paper-based archive in the first place.
Modern electronic validation solutions are browser-based and built in the cloud. With information digitized, indexed and centralized, document searching is near-instant with a few simple keywords. These keywords are matched to the document’s title, its rich metadata set, and its actual full-text content. There is also the added benefit of mobility. Users can search and retrieve documents from multiple sites without being physically on-site.
9. Pain-free inspections and audits
A four-drawer filing cabinet filled with paper files can easily weigh more than 400 pounds or 180 kg.
Data mobility in a paper-based validation system quickly becomes a massive burden. However, in the case of an FDA or ISO audit, this doesn’t matter. The necessary data must be handed over, including all relevant notes and tracking information regarding test results, revisions, changes, reviews, approvals and more.
If it’s all restricted to paper, assembling and transporting those records is a ridiculous, troublesome cost. Regulatory auditors are not interested in hearing about an organization’s delivery problems. They just want the data.
On the other hand, with a paperless/electronic solution, users are readily equipped with a complete archive of digitized records, tightly organized, with fully automated, systemgenerated audit trails. These tracked, tamper-proof audit trails allow any organization to consistently maintain and fulfill the requirements of both EU and U.S. Good Manufacturing Practice (GMP), including EU Annex 11 and FDA 21 CFR Part 11. When audits occur, your data is already organized, stored and readily available for the auditors with minimal prep work.
10. Flexible system scalability
An electronic validation solution offers scalability that doesn’t exist in a paper-based system.
Adding a new facility to an electronic validation system should be as easy as having the Admin set up a new site on the application. For cloud-based validation solutions, there is no need to set up any additional infrastructure at a new facility.
This can reduce the costs of any company's expansion. A valid paperless validation solution should be able to scale up (or, in some cases, down) based on an organization's growing/changing needs.
Pain-Free Paperless Validation
Paper-based validation systems are more time-consuming, less accurate, and terribly inefficient in compared to the electronic validation solutions on the market today.
The use of paper-based systems are more than troublesome; they are outdated and unnecessary. With an electronic validation lifecycle management system, you can:
-
Enforce standard validation processes through frameworks
-
Document consistency through template management
-
Associate validation tasks with change requests and risk assessments
-
Manage and assess risk at the system and functional level
-
Generate ad hoc reports to build any number of reports quickly and easily
-
Access validation data in real-time
Since 2005, ValGenesis has led the way in developing and improving electronic/paperless validation solutions for life sciences organizations. Our Validation Lifecycle Management System (VLMS) was the first 100% electronic enterprise software solution capable of managing the corporate validation process. The ValGenesis VLMS offers our clients a system of records and standards for all types of validation processes, and it has been in use with several well-known and globallyregulated life science companies for many years.
Why a purpose-built validation lifecycle management system?
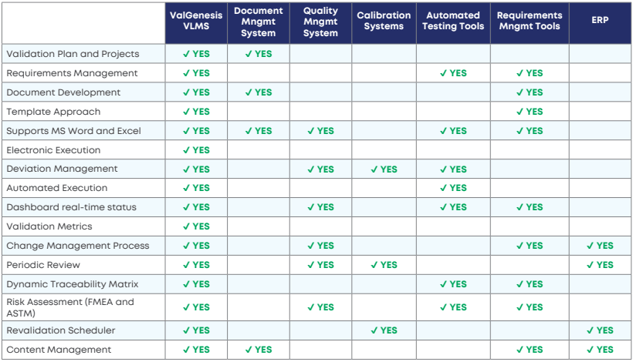
ValGenesis is the creator of an innovative software platform that serves as a foundation for managing compliance-based validation activities in life sciences companies.