Faster Validation.
Smarter Execution.
Always Audit-Ready.
ValGenesis iVal™ accelerates end-to-end validation by automating authoring, execution, and traceability for faster cycles, reduced effort, and greater confidence in every deliverable.
Global Adoption. Powerful Results.
Join the industry leaders who are replacing disconnected documents and silos with a unified validation lifecycle approach.













Customer Testimonials

We chose ValGenesis because they were a known and trusted vendor. We had limited time and resources, and we knew the quicker we could implement the solution, the sooner we’d start realizing value from it.
Joshua Pelina
Validation Engineer

Working with ValGenesis was great. They helped us align the system with our new CSV program and uncover opportunities for process improvements and efficiency gains to maximize value.
Jeff Onis
Global IT Data Integrity Manager
Impact Metrics
Audit readiness with traceable, secure digital workflows
Reduction in validation related observations
Reduction in time to complete testing activities
Reduction in validation cycle time through AI-powered content creation and execution
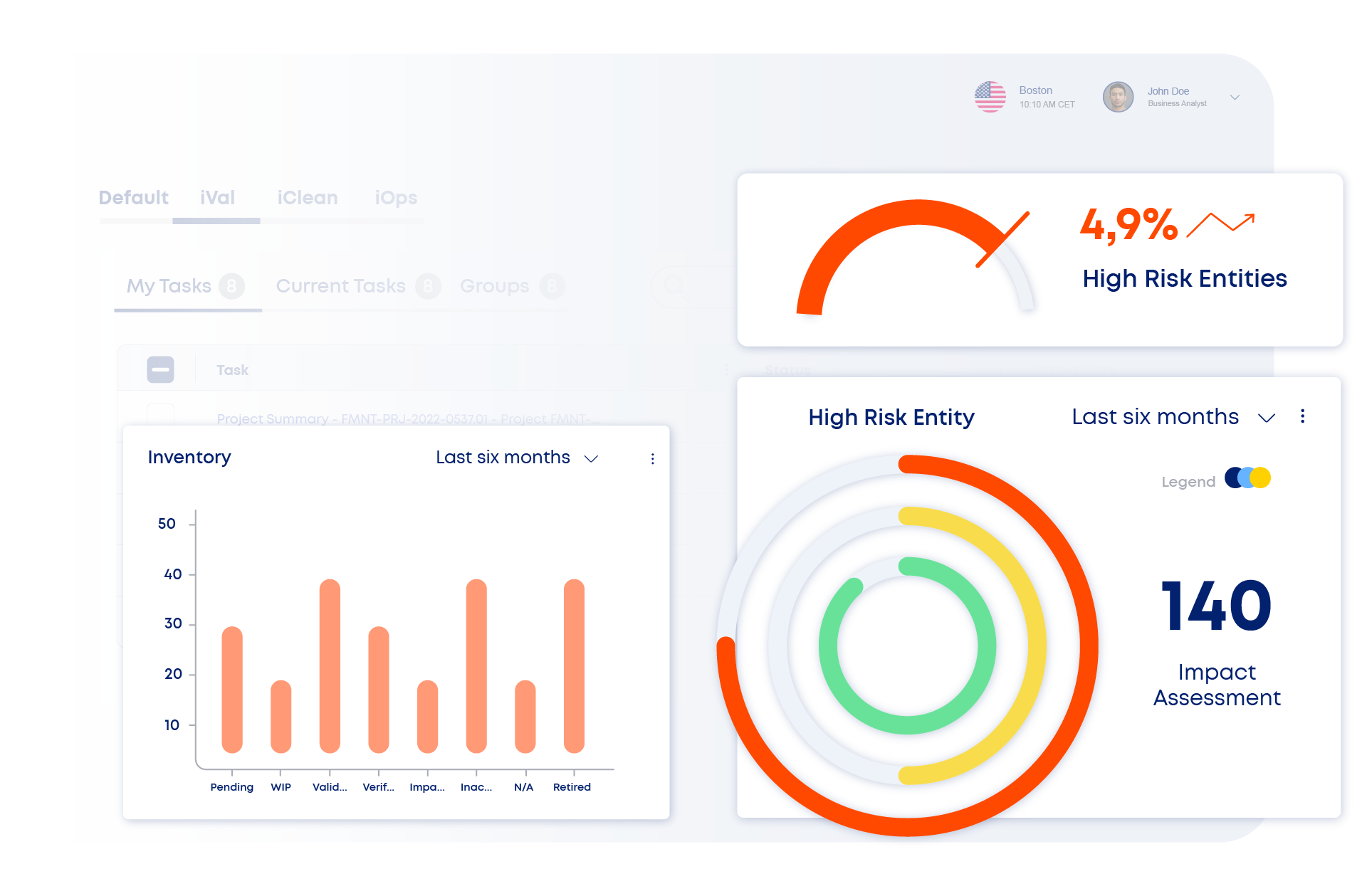
Discover How iVal Works as Part of Our Validation Lifecycle Suite
iVal is the AI-enabled backbone of your digital, automated validation tool—centralizing planning, test authoring, execution, and oversight in one intelligent system. AI-powered tools generate validation content, automate test execution, flag anomalies in real time, and analyze content to detect gaps or GDP issues based on approved SOPs. Integrated risk assessments, automated traceability matrices, and change impact tracking ensure every decision is data-driven and audit-ready.
iVal supports CSA, CQV, and CSV with offline execution, automated deviation handling, and full lifecycle traceability—accelerating time to market and reducing time to production volume, all while maintaining confident, audit-ready compliance.
Overview
Accelerate validation with less effort
Shorten cycles by up to 80% through paperless validation and AI-powered execution.
Stay compliant, anywhere
Offline execution with full traceability ensures continuous readiness.
Scale globally with control
Standardize across sites with seamless integration and flexible workflows.
Resources

Best Practices in Commissioning and Qualification
Use this C&Q best-practices guide to reduce rework and speed release decisions across ...
Read
The Impact of Artificial Intelligence on CQV
Speed CQV documentation, execution, and quality decisions with AI while meeting regulatory ...
Read
The Shift from CSV to CSA
Explains why life sciences companies should move from CSV to CSA, with FDA draft guidance context, ...
Read.png)
Regulatory Scrutiny: Why a Paperless Approach is a Non-Negotiable in CQV
Meet tougher CQV inspections with paperless validation. See the regulatory drivers and readiness ...
ReadWebinars

Digital CQV and Pharma 4.0: Paving the Way for Future Success
Imagine a future where manual paperwork and administrative tasks in commissioning, qualification ...
Watch
Boost Efficiency and Quality with CSA and Modern Software Tools
In this webinar, we will explore the transformative power of Computer Software Assurance (CSA). By ...
Watch
Best Practices in Commissioning and Qualification
Commissioning and qualification (C&Q) ensure that facilities, systems, utilities, and equipment ...
Watch
CQV: From a Painful Manual Approach to a Smooth Digital Process
Tired of drowning in manual work and inefficiencies with your CQV plan? Join our upcoming webinar ...
Watch
Challenges and Solutions: A Deep Dive Into Digital CQV
With rapid advancements in technology and increasing regulatory expectations, the pharmaceutical ...
WatchVideos

AI-Powered CQV That’s Still Audit-Ready: What "Good" Looks Like
Learn how to keep AI-powered CQV audit-ready with governed workflows and full traceability—watch ...
Watch
ValGenesis iVal™: Faster Validation. Smarter Execution. Always Audit-Ready.
Cut validation cycle time up to 80% with AI-assisted workflows and traceability. Watch the video to ...
Watch
Advancing CQV: The Role of AI in Modern Validation Processes
Recover lost validation time, reduce protocol deviations, and speed CQV cycles with AI-driven ...
WatchFrequently Asked Questions
Go Paperless. Go Faster.
Book a demo and watch iVal put digital validation on fast‑forward.