
Published on March 14, 2024
Reading time: -- minutes
Last updated on April 13, 2026


Microsoft Excel remains the predominant tool for risk management across the life sciences industry. Spreadsheets are inherently limited, but their adaptability, user-friendly interface, and cost-effectiveness have rendered them the favorite choice among life sciences risk management professionals.
But spreadsheets are not enough. If you read this blog post, you’ll understand why relying on Excel is insufficient to navigate risk management in the pharmaceutical industry effectively.
The year 2020 kicked off a sequence of events that impacted most industries: a pandemic, disruptions in supply chains, geopolitical instabilities, and fluctuating inflation and interest rates.
The levels of risk exposure are higher than ever. Regulators now demand that you take a proactive stance toward risk management as specified in the revisions of ICH Q9 (Guideline on Quality Risk Management) and GMP Annex 1. In light of these developments, re-evaluating the risk management proficiency and tangible benefits derived from risk registries has become evident and urgent.
To comply with these recent changes, an effective risk management system should:
As you can see, merely identifying and documenting risks falls short of addressing regulatory expectations for effective risk management. There is also a pressing need for heightened accountability, which calls for regular senior leadership engagement with identified risks and diligent pursuit of remedial actions. Moreover, risk management is no longer the purview of a specific team or individual but a concern across the organization. As such, we need a structured methodology and procedural framework to ensure companywide risk management. Spreadsheets do not allow that.
In fact, if you made a list of all the things a risk management tool should do that Excel cannot, you would end up with something close to this:
Spreadsheets are tedious and time-consuming to maintain. As risk management activities expand within the company for different products at different stages of the lifecycle, your team may find itself working on outdated versions of documents or having to search multiple sources to retrieve the information they need.
Digitalizing your risk management process under one specialized platform can have a major impact on your team’s performance. It can address risk maturity, cultural alignment, and organizational capability while also enforcing accountability, procedural consistency, action tracking, and report streamlining.
While Excel may offer convenience and accessibility, it has become increasingly inadequate for effectively managing the diverse and evolving risks within the pharmaceutical industry.
Life sciences companies that want to shield themselves from manufacturing and auditing vulnerabilities — which have very negative business impacts — should look for risk management software that is contemporary, user-friendly, and cost-effective.
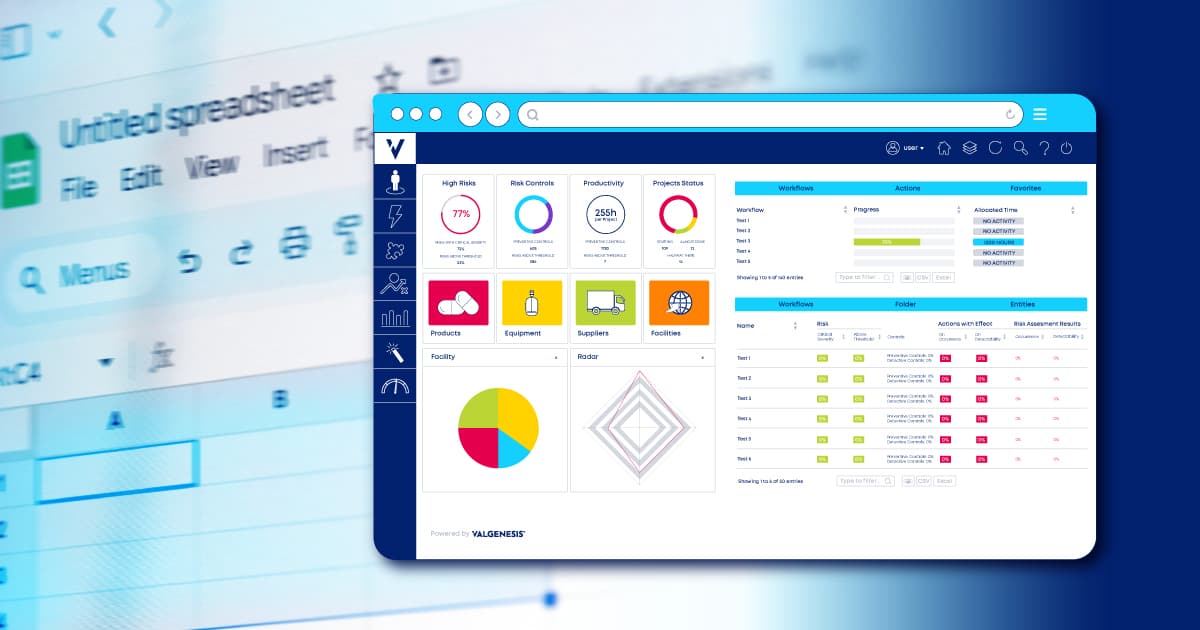
ValGenesis iRisk is the most advanced QRM platform for the life sciences. It is built on industry best practices and designed by subject matter experts with real-world expertise.
The software unifies all risk-related activities, allowing you to identify, quantify, and prioritize risks in one powerful platform. It also allows you to keep track of risk status with user-friendly dashboards. Most importantly, it eliminates the shortcomings and vulnerabilities that using Microsoft Excel for risk management generates.
Learn why Quality by Design (QbD) often fails post-approval and how pharma leaders can ensure decision traceability for continuous improvement and success.
By Sofia Santos

Improve the efficacy of your risk management process with these 6 strategies inspired by the ICH Q9 quality risk management guideline.
By Margarida Ventura

Discover why transitioning to a paperless approach in CQV is essential to meet regulatory expectations and ensure data integrity in pharma manufacturing.
By Sweta Shah