Risk-based.
Justified Limits.
Digital Cleaning Validation.
ValGenesis iClean™ streamlines cleaning validation with global standardization, automated oversight, and scientific justification—ensuring audit readiness across all sites.
Impact Metrics
digital documentation and traceability with automated audit trails
faster cleaning validation cycle time through automation
Discover How iClean Works as Part of Our Validation Lifecycle Suite
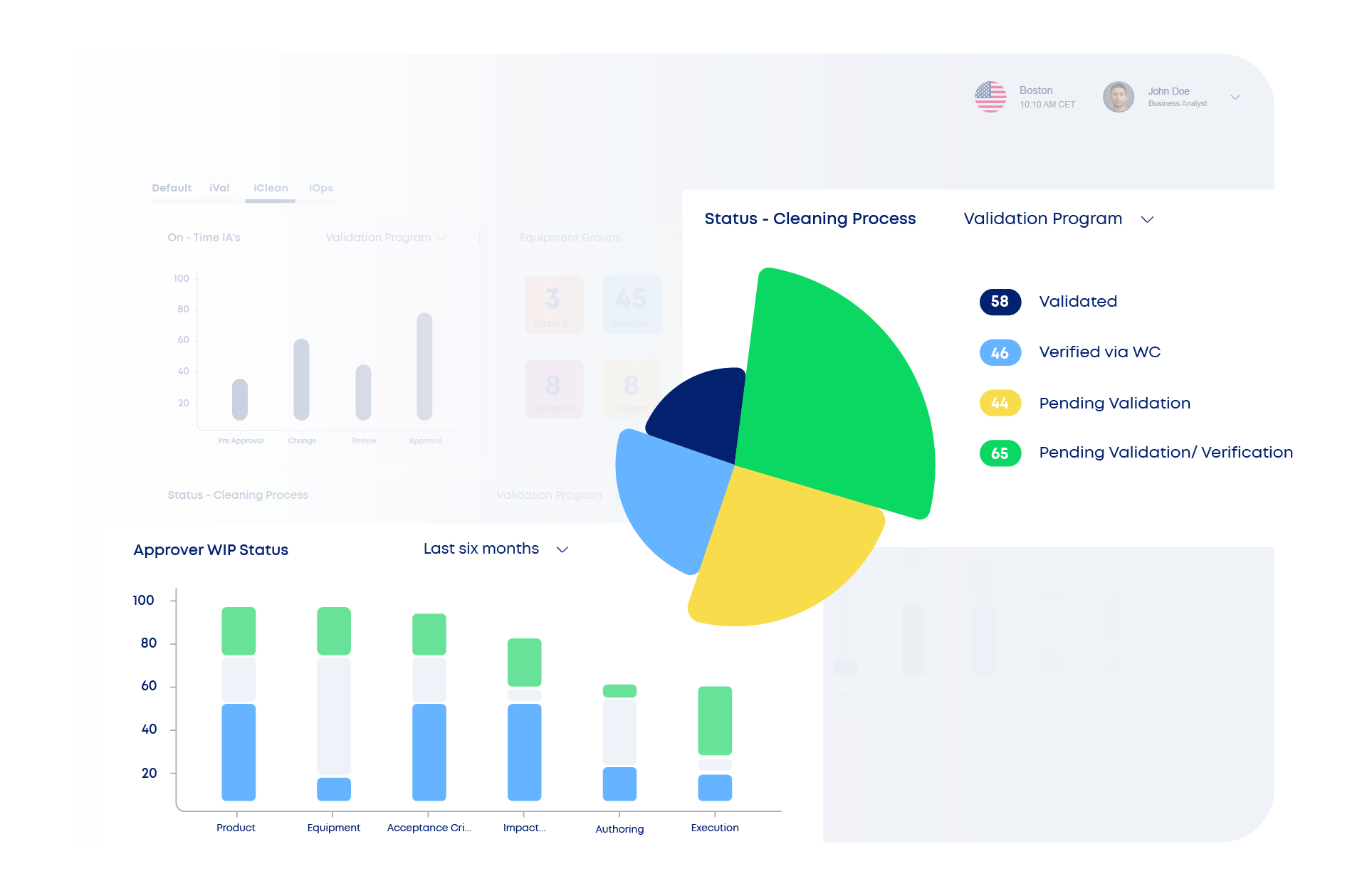
iClean digitalizes the entire cleaning validation lifecycle—from risk-based planning and MACO determination to execution and ongoing requalification. It automates calculations, integrates ADE/NOAEL-based scientific data, and uses 2D/3D equipment mapping to guide the sampling strategy.
With business-rule-driven decision trees, built-in residual limit enforcement, and a centralized method and agent repository, iClean helps teams reduce manual work, eliminate errors, and maintain global consistency. Its change control and requalification workflows ensure sustained compliance—even as processes evolve.
Overview
Accelerate compliance, reduce risk
Automate MACO calculations, validation cycles, and documentation for faster, inspection-ready delivery.
Standardize globally, flex locally
Centralize cleaning strategies while adapting validation protocols to site-specific and equipment-specific requirements.
Gain real-time oversight
Visualize status, risks, and readiness across sites through built-in analytics and intelligent dashboards.
Resources

The Future of Cleaning Validation
Modernize cleaning validation with digital CPV, better documentation, and early deviation ...
Read
Unlocking Operational Capacity with Fully Digitalized Cleaning Validation
Cut cleaning-validation downtime and audit stress with a fully digital approach. Read the Industry ...
ReadWebinars

Digital Cleaning Validation Lifecycle
Cleaning validation is a complex process. Using manual, paper- or Excel-based processes creates a ...
Watch
Cleaning Validation Programmes: Establishing a Compliance-Led Framework
In the pharmaceutical industry, establishing an effective Cleaning Validation (CV) program is ...
Watch
Common FDA 483 Cleaning Validation Observations and How to Avoid Them
The pharmaceutical industry continues to face challenges in maintaining proper cleaning regimens ...
Watch
Manufacturing Bottlenecks: Why Analog Cleaning Validation Programs Slow Down Your Entire Operation
Cleaning validation programs are complex to plan and deliver due to the multitude of regulatory, ...
WatchVideos

How Digitalizing Cleaning Validation Leads to Shorter Timelines and Fewer Errors
Speed up cleaning validation with a digital, risk‑based workflow. See how teams cut cycle times by ...
Watch
ValGenesis iClean™: Risk-based. Justified Limits. Digital Cleaning Validation.
Modernize cleaning validation with risk-based workflows, justified limits, and centralized ...
Watch
3 Ways Old-School Cleaning Validation Delays Your Production Schedule
Stop cleaning-validation changeover delays. Learn the 3 old-school blockers and how digital, ...
WatchFrequently Asked Questions
Do you need help with something or have questions about any features?
Ready for Smarter Cleaning Validation?
Simplify cleaning validation. Ensure audit readiness. Get started with ValGenesis iClean™.