Is Analytical QbD the Future of Method Development?
By now, we all know that Quality by Design (QbD) is essential to deliver safe and effective medicines. Because of that, the Pharma Industry is trying to adapt this methodology to Analytic Methods development. And it’s trying to do that by building quality into the design of an analytical test method.
In this blog post, we’ll give you an overview about what is Analytical QbD (AQbD) and answer this one question: is Analytical QbD the future of method development?
Let’s Start With Some Context!
The lack of regulations has limited the development of Analytical QbD. It happens because there are no specific guidelines to show how to apply these concepts. However, with the advent of ICH Q14 approval, it will be possible to have a clear view of the concepts of Quality by Design to analytical methods and their application.
This new guideline will be important to the new concept. Why is that you ask? Because it proposes the harmonization of scientific approaches and provides principles for the development of analytical processes.
But, After All, What is the Analytical QbD?
As the name says, the Analytical QbD is an extension of QbD. However, it can offer you a systematic and robust approach to the development of analytical procedures involving all the stages of a product’s lifecycle. Many say that Analytical QbD is the key to method accuracy and precision, and we couldn’t agree more. Among the many benefits, we want to focus on:
- It helps identify and minimize the variability sources that cause poor method robustness.
- It ensures that the method meets the intended performance requirements through the lifecycle.
Overall, AQbD helps you in developing a robust and cost-effective analytical method. That can be applicable throughout the lifecycle of the product, to facilitate regulatory flexibility in analytical methods.
And What Can You Expect From Analytical QbD?
First of all, implementing Analytical QbD in your manufacturing process as a part of your control strategy will help you predetermine performance and product quality.
Also, having a more robust analytical data is becoming a need for pharmaceutical development. So, we believe that this will intensify the rigor in analytical method development. And, more than that, will leverage the industry view that analytical QbD is indeed necessary.
In fact, analytical methods are indicators of process quality, product, and robustness throughout the lifecycle.
One-Factor-At-Time (OFAT) vs AQbD
Usually, the analytical methods are based on one factor at a time (OFAT). This traditional approach optimizes only one parameter for the expected results while many others remain constant. However, when it comes to analytical method development, this strategy has high risks and it generally requires a revalidation protocol after any method change or transfer.
AQbD otherwise explores scientific understanding in method implementation. Just have a look at the table below and check the differences between the conventional approach and the analytical QbD:
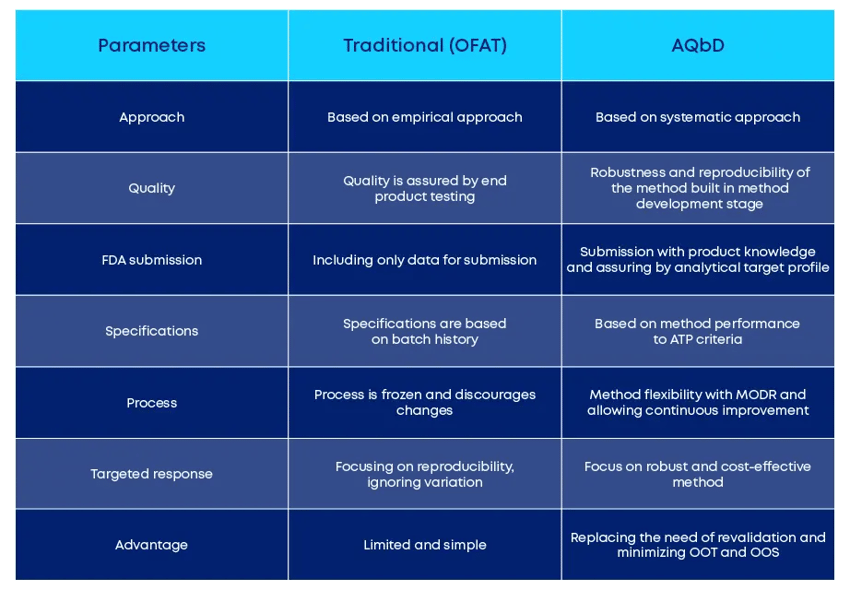
Figure 1 – Differences between the traditional (OFAT) vs. AQbD approach
So, Why is it Important to Implement an AQbD in Your Method Development?
More than a development of more robust methods, the application of a QbD Approach to analytical methods will optimize the methods of your risk assessments as well as the strategies of control assignments. Also, it can provide you with the ability to assess robustness throughout the development process and perform quality risk management when a method is transferred.
By applying the AQbD, your methods will be developed using a risk-based approach guaranteeing quality assurance in all your processes. We are sure that this represents an advantageous alternative to the traditional approaches!
References
R. Peramn, K. Bhadraya, Y. P. Reddy. “Analytical Quality by Design: A Tool for Regulatory Flexibility and Robust Analytics”. International Journal of Analytical Chemistry. 2015
View at: Publisher Site
Related Blog Posts

Does Quality Culture Affect pQMS Outcomes? Absolutely.
This post examines the crucial impact quality culture has on individuals, organizations, and processes; it offers tips for improving it in your company.
By Dr. Ajaz Hussain

Medical Cannabis: the QRM Practices that Improve Your Business
A blog post where we give you some tips on how Quality Risk Management can help you improve your Medical Cannabis business
By Manuel Silva

Cell and Gene Therapies: Risk Management Processes
In this blog post, we talk about how can you implement Risk Management Processes for Cell and Gene Therapies to every aspect of your product.
By Manuel Silva